Incandescent (glowing) solids such as tungsten filaments in incandescent lights also give off light that contains all wavelengths of visible light. (credit: modification of work by American Society for Testing and Materials (ASTM) Terrestrial Reference Spectra for Photovoltaic Performance Evaluation) The unabsorbed sunlight at the top of the atmosphere has a distribution that approximately matches the theoretical distribution of a blackbody at 5250 ☌, represented by the blue curve. wavelength) of sunlight reaches the Earth's atmosphere as UV light, visible light, and IR light. The spectral distribution (light intensity vs. As can be seen in Figure 9.1, sunlight also contains UV light (shorter wavelengths) and IR light (longer wavelengths) that can be detected using instruments but that are invisible to the human eye. You can see all the visible wavelengths of light present in sunlight by using a prism to separate them. Most of the light generated from stars (including our sun) is produced in this fashion. Photons produced in this manner have a range of energies, and thereby produce a continuous spectrum in which an unbroken series of wavelengths is present. When solids, liquids, or condensed gases are heated sufficiently, they radiate some of the excess energy as light. Using the Bohr model, we can calculate the energy of an electron and the radius of its orbit in any one-electron system.Īnother paradox within the classical electromagnetic theory that scientists in the late nineteenth century struggled with concerned the light emitted from atoms and molecules. 
To move an electron from a stable orbit to a more excited one, a photon of energy must be absorbed. When an electron moves from a higher-energy orbit to a more stable one, energy is emitted in the form of a photon. 
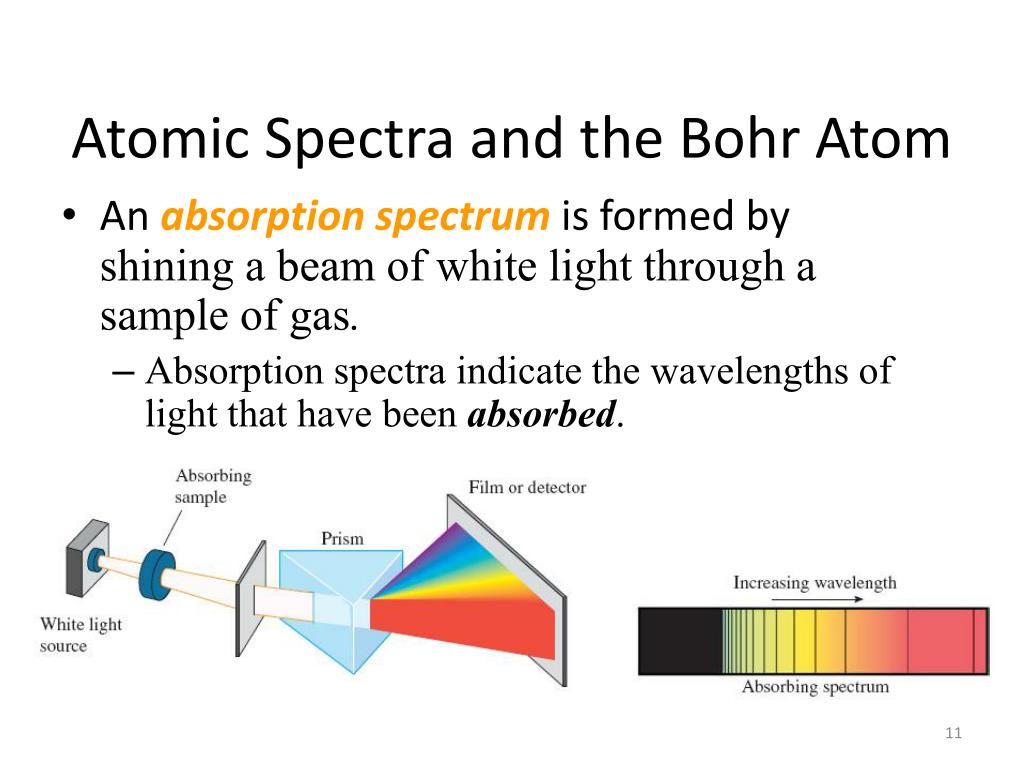
Transitions between these allowed orbits result in the absorption or emission of photons. He postulated that the electron was restricted to certain orbits characterized by discrete energies. Bohr described the hydrogen atom in terms of an electron moving in a circular orbit about a nucleus. The Bohr model of the hydrogen atom explains the connection between the quantization of photons and the quantized emission from atoms. The resolution of these paradoxes ultimately led to quantum theories that superseded the classical theories.īohr incorporated Planck’s and Einstein’s quantization ideas into a model of the hydrogen atom that resolved the paradox of atom stability and discrete spectra. Three historically important paradoxes from the late 19th and early 20th centuries that could not be explained within the existing framework of classical mechanics and classical electromagnetism were the blackbody problem, the photoelectric effect, and the discrete spectra of atoms. This line spectrum was simple enough that an empirical formula called the Rydberg formula could be derived from the spectrum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
